Shape-shifting chemical is key to new solar battery
It can absorb and hold energy until needed hours to months later

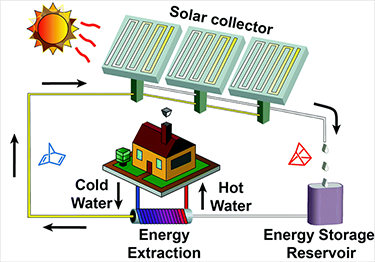
Kasper Moth-Poulsen shows off the solar collector his team used to test its liquid battery.
Johan Bodell/Chalmers University of Technology
What powers the device you’re using? Electricity, obviously. But where did that come from? Two thirds of the electricity used in the United States comes from power plants fueled by fossil fuels — coal, oil or natural gas. Solar energy produces just 1.3 percent of the electricity. Yet energy from the sun could easily power our every need if it could be stored for use when the sun doesn’t shine (such as at night). Researchers in Sweden now think they might have a way to do just that.
As a chemical engineer, Kasper Moth-Poulsen uses chemistry and physics to design solutions to problems. He works at Chalmers University of Technology in Gothenburg, Sweden. He teamed up with other researchers in Sweden and Spain to tackle the problem of storing energy from the sun. Their solution: Store that energy inside the bonds of molecules that have been suspended in a liquid.
Molecules consist of two or more atoms. Those atoms share electrons through bonds that hold them together.
Different types of molecules have distinct 3-D shapes. For example, methane is shaped like a three-sided pyramid called a tetrahedron (Teh-tra-HE-drun). Other molecules have different shapes. Adding energy to a molecule can alter its shape. New bonds may now form between its atoms — ones that may hold different amounts of energy. When a molecule later absorbs energy, that energy can become trapped within those new bonds.
That’s the key to the new solar-energy battery.
Using bonds inside a molecule to store solar energy isn’t new. Moth-Poulsen’s group had been working on that for years. But the molecules it initially worked with contained a rare and pricey metal called ruthenium (Roo-THEE-nee-um). The researchers needed a less costly alternative.
For inspiration, they turned to work by other chemists. They soon found a promising candidate called norbornadiene (Nor-BORN-uh-DY-een). It is made mostly of carbon and hydrogen, atoms that are found in all living things. That means this molecule should be cheap and easy to make.
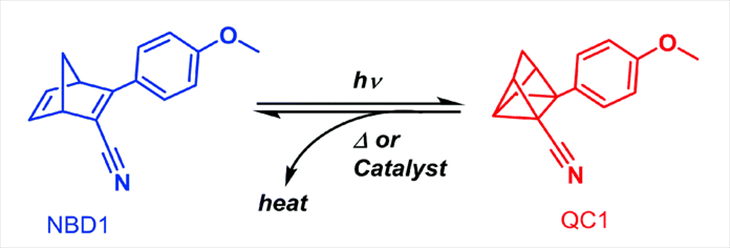
Yet there was still one problem. This chemical could absorb only ultraviolet (UV) light — a small part of the sun’s light. To make this molecule more useful, the researchers tweaked it in such a way that it would absorb more wavelengths (colors) of sunlight. That innovation sounds easy. In fact, it took them seven years to pull off. Now their molecule can absorb energy not only from UV, but also from blue and green light.
One end of the molecule reacts to this light. As it absorbs this energy, the molecule snaps into a new shape. New bonds between its atoms trap that energy. And they hold it tight, even after the molecule cools to room temperature.
But storing energy isn’t useful unless you can release that energy when you need it. So Moth-Poulsen’s team found a way to get its molecule to release the stored energy as heat. Researchers pass the liquid over a type of salt that acts as a catalyst. (Catalysts are materials that speed up chemical reactions. They’re not used up in the reactions, but nudge reactions along.) The salt causes the molecule to change back into its original shape. When it does so, the molecule releases the energy stored in its bonds. That raises the temperature of the liquid by 63.4 degrees Celsius (114 degrees Fahrenheit) — enough to heat a home.
The team published its findings in the January issue of Energy & Environmental Science.
Solar-powered future
A liquid battery made with these molecules can store solar energy for days, months or even years, Moth-Poulsen says. So energy absorbed during long summer days can be held for use at night or during the winter, when days are short.
The team has tested its system in a rooftop experiment at their lab in Sweden. The system works well — but not yet well enough to put in every home. First, the team needs to increase how much of the sun’s energy the molecule can absorb. “We are aiming at reaching 5 to 10 percent” of that energy, Moth-Poulsen says.
Storing more energy in the molecule’s bonds means these could later release more heat. And while the system doesn’t make electricity, the heat it releases could be used to drive a turbine that does, Moth-Poulsen says. One day, such a system might both heat and power buildings without any connection to outside sources of electric power. Those buildings also could stay warm without a need for energy from fossil fuels.
“We have discovered some new tricks recently,” Moth-Poulsen says. He hopes these will help the home-heating system work even better. That should increase its affordability and attractiveness.
Jeffrey Grossman finds the new data exciting. This study, he explains, “demonstrates real world use of this technology.” Grossman is a materials scientist at the Massachusetts Institute of Technology in Cambridge. He was not involved with the study.
Deepa Khushalani was not either. She’s a chemist and materials scientist at the Tata Institute of Fundamental Research in Mumbai, India. Right now, Khushalani is less excited about the new technology’s prospects for making electricity. To drive a turbine or other engine, she notes, the molecule must release enough heat to turn water into steam. That means the system would need to heat water to more than 100° Celsius (212° Fahrenheit). Batteries that store electricity are a more practical way to harness the sun’s energy, she suspects.
But Moth-Poulsen plans to get the extra heat needed from the new energy-storage molecule. His team is working to make it absorb energy from yellow and orange light, too. Heating and cooling accounts for almost half of the energy used in the European Union. “If [the new technology] can just make a small impact on this, it will make a huge difference,” he says.
This is one in a series presenting news on technology and innovation, made possible with generous support from the Lemelson Foundation.







