Scientists Say: Ion
This term describes atoms or molecules with an electrical charge
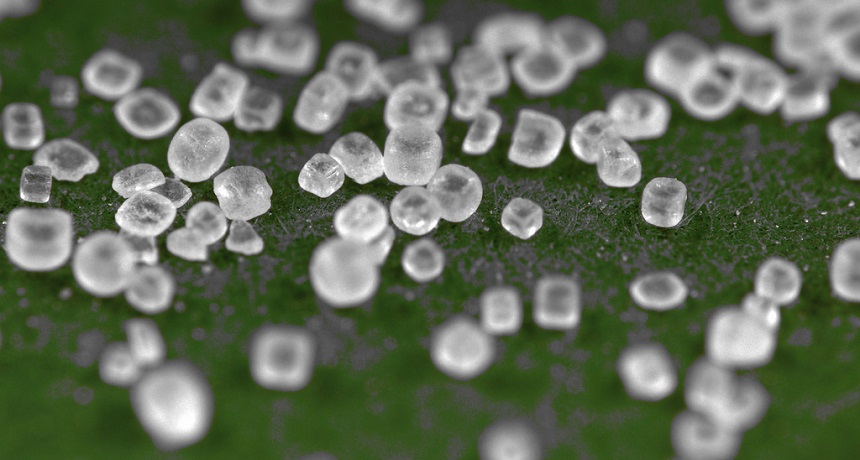
This table salt is a chemical made of two different ions. Chloride is negatively charged and sodium is positively charged.
Kevin Dooley/Flickr (CC BY 2.0)
Come explore with us!
This term describes atoms or molecules with an electrical charge

This table salt is a chemical made of two different ions. Chloride is negatively charged and sodium is positively charged.
Kevin Dooley/Flickr (CC BY 2.0)
An ion is an atom or molecule that has an electrical charge. The charge can be positive or negative. Atoms are made of protons, neutrons and electrons. The protons have a positive charge and the electrons have a negative charge. Neutrons have no charge at all. When there are equal numbers of protons and electrons, the charges cancel each other out. But when there are more protons than electrons, the molecule or atom will be an ion with a positive charge. When electrons outnumber the protons, the resulting ion is negatively charged.
Positively and negatively charged ions can bond together to create a neutral chemical, such as a salt.
Follow Eureka! Lab on Twitter
atom The basic unit of a chemical element. Atoms are made up of a dense nucleus that contains positively charged protons and neutrally charged neutrons. The nucleus is orbited by a cloud of negatively charged electrons.
chemical bonds Attractive forces between atoms that are strong enough to make the linked elements function as a single unit. Some of the attractive forces are weak, some are very strong. All bonds appear to link atoms through a sharing of — or an attempt to share — electrons.
chlorine A chemical element with the scientific symbol Cl. It is sometimes used to clean water. Compounds that contain chlorine are called chlorides.
electric charge The physical property responsible for electric force; it can be negative or positive.
electron A negatively charged particle, usually found orbiting the outer regions of an atom; also, the carrier of electricity within solids.
ion An atom or molecule with an electric charge due to the loss or gain of one or more electrons.
molecule An electrically neutral group of atoms that represents the smallest possible amount of a chemical compound. Molecules can be made of single types of atoms or of different types. For example, the oxygen in the air is made of two oxygen atoms (O2), but water is made of two hydrogen atoms and one oxygen atom (H2O).
neutron A subatomic particle carrying no electric charge that is one of the basic pieces of matter. Neutrons belong to the family of particles known as hadrons.
proton A subatomic particle that is one of the basic building blocks of the atoms that make up matter. Protons belong to the family of particles known as hadrons.
sodium A soft, silvery metallic element that will interact explosively when added to water. It is also a basic building block of table salt (a molecule of which consists of one atom of sodium and one atom of chlorine: NaCl).