Gene editing creates mice with no mom
The first rodents with two biological dads only survived a few days
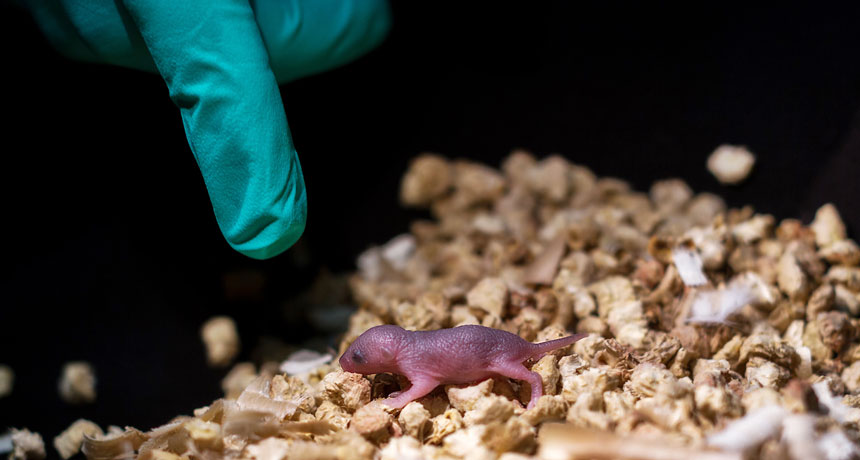
This mouse pup has two biological fathers and no mother. It was made in a lab using stem cells made from two male mice. Such pups, including this one, lived a few days at most.
LEYUN WANG
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
For the first time, mice have been born with no moms. Researchers created them from genetic material retrieved from two dads. Alas, the pups did not live long.
Similar mice created with no dads, fared better. Those that were born tended to survive into adulthood. Some even gave birth to their own pups.
Scientists had to edit the animals’ genes for either type of moms-only or dads-only pups to be born. That’s why this unusual reproduction took place in a lab.
Researchers started by turning skin cells from male mice into stem cells. Such cells can later become any type of cell. Biologists turned the stem cells into a type similar to those that become eggs or sperm. Then they injected these pre-egg cells with sperm from another male. These grew into embryos. Finally, the scientists implanted the embryos into female mice. Although those females gave birth to the mouse pups, they were not genetically related.
The researchers described their novel findings October 11 in Cell Stem Cell.
Pups without pops
The researchers wanted to learn why mammals need two parents of opposite sexes to reproduce.
Sperm from one parent usually are needed to fertilize an egg from the other parent. But certain non-mammals can sometimes reproduce with only one parent. These include turkeys, snakes and sharks, notes study coauthor Qi Zhou. Zhou works at the Chinese Academy of Sciences in Beijing. Females of those species can sometimes make young from an unfertilized egg. This process is called parthenogenesis (PAR-then-oh-JEN-eh-sis).
Researchers had made zebrafish before that had only the DNA of one father. But until now, no one had gotten mammals to reproduce without moms.

For their new study, scientists started by making mouse pups with two mothers. These pups were smaller than usual and born with some abnormalities.
The problem: Some genes that the pups inherited from their mothers were “marked.” The marks are molecules called methyl groups. They are attached to DNA near a gene. This type of epigenetic marking — also known as imprinting — can make genes more or less active.
To avoid this, the researchers used a type of molecular “scissors.” It snipped out three imprinted DNA regions near important genes. These scissors are known as CRISPR/Cas9. The scientists edited the DNA of female pre-sperm stem cells in this way. Then they injected the edited cells into eggs from other female mice to make embryos.
The researchers implanted 210 of these embryos in female mice. About 14 percent of them survived to birth. These pups grew normally and became adults. These dad-free females later gave birth to their own pups. This result matched what the researchers had seen before.
Mice minus moms
Making embryos from the stem cells of two male mice wasn’t nearly as successful.
Male-only reproduction is called androgenesis (An-droh-JEN-eh-sis).
To make embryos from the stem cells of two dads, the researchers had to snip out twice as many imprinted pieces of DNA — six, not three.
Then they implanted 1,023 of these embryos into female mice. Out of all these embryos, only 1.2 percent survived to birth. Those pups were twice the normal size and soon died. Cutting out a seventh imprinted DNA region produced pups of normal size. But only two of these lived more than 48 hours. Neither lived to become an adult.
(The researchers point out that even in their study, they couldn’t get away with having no females at all. After all, females had to carry the embryos with two dads in their wombs. For that reason, it would be extremely hard for males to have babies on their own in the wild.)
This work is an important first step in understanding the role of imprinted regions in fetal development, says B. Duygu Özpolat. She’s a developmental biologist at the Marine Biological Laboratory in Woods Hole, Mass. She was not involved in the new work.
Imprinting errors are important to understand. They underlie some human birth defects and genetic conditions. So knowing how the process works might help scientists one day correct those defects, Özpolat says.
For instance, the Chinese team notes that one of the imprinted regions snipped out of the two-mom mice is similar to a region in people. When a human gets an extra copy of that piece of DNA, they develop what’s known as Silver-Russell syndrome. Fetuses and babies with the extra imprinted bit don’t grow normally. In the future, snipping out the extra imprinted DNA might lead to healthy babies, these researchers say.
Making mammals from parents of the same sex might also offer some help to certain endangered species, Özpolat says. Some species are represented only by animals of one sex. For instance, the last male northern white rhinoceros died earlier this year, leaving just two females. Gene editing might help researchers bring white rhinos back by making all-female populations from stem cells grown in labs.
Although possible, Özpolat says, “It might be too expensive, and might not work for every species. But it’s something.”
Some human same-sex couples hope to have a biological baby together. But, Özpolat says, “that’s the far future.” Right now, it remains just a dream.
Zhou adds that it could be too dangerous to try this technique in people. There’s no guarantee that the imprinted regions involved in mouse reproduction are the same ones involved in people. In fact, he adds, using this technique with people “is not one of our goals.”







