Living Mysteries: Meet Earth’s simplest animal
Tiny Trichoplax, discovered in a fish tank, provides a window into the origin of animals

Dickinsonia was one of the first animals on Earth. It lived on the ocean floor 550 million to 560 million years ago. It looked like a giant version of the modern-day Trichoplax, the simplest animal now alive.
John Sibbick
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
By Douglas Fox
Living Mysteries launches as an occasional series on organisms that represent evolutionary curiosities.
Franz Eilhard Schulze had a laboratory full of beautiful sea creatures. In the 1880s, he was one of the world’s top experts on ocean sponges. He found many new species and filled saltwater aquariums at the University of Graz in Austria with these simple sea animals. They were striking — brightly colored with exotic shapes. Some looked like flower vases. Others resembled miniature castles with pointy towers.
But today, Schulze is best remembered for something very different — a drab little animal no larger than a sesame seed.
He discovered it one day by pure accident. It was hiding in one of his fish tanks. Creeping along the inside of the glass, it was dining on the green algae that grew there. Schulze named it Trichoplax adhaerens (TRY-koh-plaks Ad-HEER-ens). That’s Latin for “hairy sticky plate” — which is about what it looks like.
To this day, Trichoplax remains the simplest animal known. It has no mouth, no stomach, no muscles, no blood and no veins. It has no front or back. It is nothing but a flat sheet of cells, thinner than paper. It is only three cells thick.
This little blob might look boring. But scientists are interested in Trichoplax precisely because it is so simple. It shows what the very first animals on Earth might have looked like, 600 million to 700 million years ago. Trichoplax is even providing hints about how simple animals later evolved more complicated bodies — with mouths, stomachs and nerves.
A hungry suction cup
At first glance, Trichoplax does not even look like an animal. Its flat body constantly changes shape as it moves. As such, it resembles a blob called an amoeba (Uh-MEE-buh). Amoebas are a type of protist, single-celled organisms that are neither plants nor animals. But when Schulze looked through his microscope in 1883, he could see several clues that Trichoplax truly was an animal.

Some amoebas are larger than this animal. But an amoeba has just one cell. In contrast, the body of a Trichoplax has at least 50,000 cells. And though this animal lacks a stomach or heart, its body is organized into different kinds of cells that perform different tasks.
This “division of labor between cell types” is a hallmark of animals, explains Bernd Schierwater. He works at the Institute for Animal Ecology and Cell Biology in Hannover, Germany. He’s a zoologist who has been studying Trichoplax for 25 years.
Cells on the underside of Trichoplax have tiny hairs called cilia (SILL-ee-uh). The animal moves by twirling these cilia like propellers. When the animal finds a patch of algae, it stops. Its flat body settles down atop the algae like a suction cup. Some special cells on the underside of this “suction cup” squirt out chemicals that break down the algae. Other cells absorb the sugars and other nutrients released from this meal.
So the animal’s entire underside works as a stomach. And since its stomach is on the outside of its body, it doesn’t need a mouth. When it finds algae, a Trichoplax just plops itself onto the food and begins to digest it.
Clues about the first animals
Schierwater believes that the first animals on Earth must have looked a lot like Trichoplax.
When those animals appeared, the oceans were already full of single-celled protists. Much as Trichoplax do, those protists swam by twirling their cilia. Some protists even formed colonies. They gathered into balls, chains or sheets made of thousands of cells. Many protists alive today also form colonies. But these colonies are not animals. They are just clumps of identical, single-celled organisms that happen to be living in harmony.
Then, 600 million to 700 million years ago, something happened. One group of ancient protists formed a new type of colony. Each member’s cell started out the same. But over time, those cells began to change. Once identical, they eventually morphed into two different types. All of the cells still contained the same DNA. They had exactly the same genes. But now the cells began chatting with each another. To do that, they released chemicals that served as messages. These told cells in different parts of the colony to do different things. Says Schierwater, this would have been the first animal.
He suspects that this first animal must have been a flat sheet, much like Trichoplax. It would have been just two cells thick. Those on the bottom let it crawl and digest food. Cells on the top did something else. Maybe they protected the animal from protists out to eat it.
It makes sense that the first animal would be flat. Just consider what the ocean looked like back then. Shallow areas of the seafloor were covered with a gooey carpet of single-celled microbes and algae. The first animal would have crept atop this “microbial mat,” Schierwater says. It would have digested the microbes and algae berneath it — just as Trichoplax does.
That first animal was probably no larger than Trichoplax. It left no fossils. But larger, similar animals evolved over time. Scientists have found fossils that look like giant versions of Trichoplax.
One, known as Dickinsonia, lived some 550 million to 560 million years ago. It was up to 1.2 meters (four feet) across. No one knows whether it would have been related to Trichoplax. It moved and ate the way Trichoplax does, crawling around and then plopping down on a meal. Like Trichoplax, it had no organs — tissues like a brain or eyes that work together to perform a particular task. But its body was a bit complex in other ways. It had front and back ends and left and right sides. Its flat body also was divided into segments, like a quilted blanket.
Mouth and butt — an animal starter kit?
For Schierwater, it is easy to imagine how such a simple animal could evolve a more complex body. Start with a plate of cells, like Trichoplax, whose stomach is its entire underside. The edges of that plate might gradually lengthen until it looked like a bowl sitting upside down. The opening of the bowl might narrow until it looked like an upside-down vase.
Story continues below image.
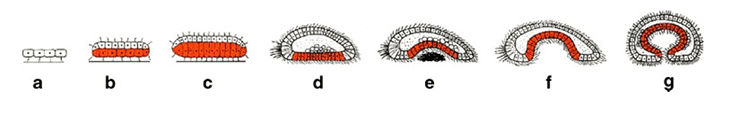
“Now you have a mouth,” says Schierwater. It’s the opening of the vase. Inside that vase is now the stomach.
When this primitive animal has digested its food, it just spits back out any unneeded remains. Some modern animals do this. Among them are jellyfish and sea anemones (Uh-NEMM-oh-nees).
Over millions of years, Schierwater suggests, this vase-shaped body stretched. As it got longer, it made a hole at each end. One hole became the mouth. The other, an anus, was where it pooped out wastes. This is the type of digestive system seen in bilaterian (By-lah-TEER-ee-an) animals. Bilaterians are a step past anemones and jellyfish on the evolutionary tree of life. They include all animals with right and left sides and front and back ends: worms, snails, insects, crabs, mice, monkeys — and, of course, us.
Deceptively simple
Schierwater’s idea that the first animal looked like Trichoplax gained some support in 2008. That year, he and 20 other scientists published its genome (JEE-noam). That’s its full string of DNA, containing all of its genes. Trichoplax might look simple on the outside. But its genes pointed to a somewhat complex inner life.
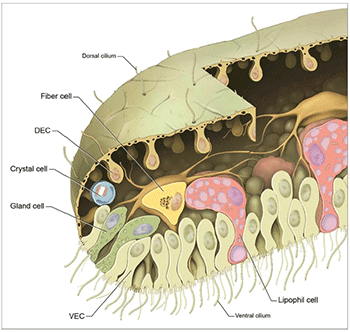
This animal has only six types of cells. For comparison, a fruit fly has 50 types. But Trichoplax boasts 11,500 genes — 78 percent as many as a fruit fly.
In fact, Trichoplax has many of the same genes that more complex animals use to shape their bodies. One gene is called brachyury (Brack-ee-YUUR-ee). It helps form the vase shape of an animal, with its stomach on the inside. Another gene helps divide the body — from front to back — into different segments. It’s known as a Hox-like gene. And as this name implies, the gene is similar to Hox genes, which shape insects into front, middle and back parts. In people, Hox genes divide the spine into 33 separate bones.
“It was a surprise” to see so many of these genes in Trichoplax, says Schierwater. This suggests that a flat, primitive animal already had many of the genetic instructions that animals would need to evolve a more complicated body. It was just using those genes for different purposes.
First nerves
Trichoplax turned out to have 10 or 20 of the genes that in more complex animals help create nerve cells. And this really grabbed the interest of biologists.
In 2014, scientists reported that Trichoplax has a few cells that act surprisingly like nerve cells. These so-called gland cells are scattered across its underside. They contain a special set of proteins known as SNARE. These proteins also show up in the nerve cells of many more complex animals. In those animals, they sit at synapses (SIN-apse-uhs). These are places where one nerve cell connects to another. The proteins’ job is to release chemical messages that move from one nerve cell to the next.
A gland cell in Trichoplax looks much like a nerve cell at a synapse. It, too, is packed with little bubbles. And just as in nerve cells, those bubbles store a kind of messenger chemical. It’s known as a neuropeptide (Nuur-oh-PEP-tyde).
Last September, scientists reported that gland cells actually control the behavior of Trichoplax. When this animal creeps over a patch of algae, these cells “taste” the algae. That informs the animal that it’s time to stop creeping.
A single gland cell can do this by releasing its neuropeptides. Those neuropeptides tell nearby cells to stop twirling their cilia. This puts putting on the brakes.
The chemicals also communicate with other nearby gland cells. They tell their neighbors to dump out their own neuropeptides. So this “stop and eat” message now spreads from cell to cell across the entire animal.
Carolyn Smith looks at Trichoplax and sees a nervous system that is just starting to evolve. In a sense, it is a nervous system without nerve cells. Trichoplax uses some of the same nerve proteins that more complex animals use. But those aren’t yet organized into specialized nerve cells. “We’re thinking of it as like a proto-nervous system,” says Smith. As early animals continued to evolve, she explains, “those cells essentially became neurons.”
Smith is a neurobiologist at the National Institutes of Health in Bethesda, Md. She and her husband, Thomas Reese, discovered the nerve-like properties of gland cells. Three months ago, they described another part of Trichoplax’s proto-nervous system. They found cells containing a kind of mineral crystal. That crystal always sinks to the bottom of the cell, whether Trichoplax is level, tilted or upside down. In this way, the animal uses these cells to “feel” which direction is up and which is down.
Creature carries snake-like venom
Trichoplax isn’t just teaching biologists about evolution, however. Scientists still are learning surprisingly basic things about how this animal lives. For one thing, it can fly! (Sort of.) Also it is deadly poisonous. And it may spend part of its life sneaking around in an entirely different shape — a disguise that scientists still haven’t recognized.
For a century after Trichoplax’s discovery, people had thought the animal could only crawl. In fact, they are skilled swimmers. And that may be how they spend much of their time, Vicki Pearse discovered. She’s a biologist, recently retired from the University of California, Santa Cruz. Back in 1989, she was traveling from one island to another in the Pacific Ocean.
She collected Trichoplax wherever she went. Afterward, she spent hours watching them under a microscope. One day, she spotted one swimming through the water “like a little flying saucer.” Once she learned to look for it, she often saw the animals swimming this way.
This wasn’t the only weird discovery that she made that year. Another time at her microscope, she watched Trichoplax being chased by a snail. She was sure she was going to see the little fellow get eaten. But as soon as the snail caught hold of Trichoplax, it pulled back as if it had touched a hot stove.
“They look completely defenseless,” she says of Trichoplax. “They’re just a little blob of tissue. They should be delicious.” But not once did she see a hungry predator actually eat one. Instead, the hunter always seemed to change its mind at the last second. “There must be something nasty about them,” Pearse thought.
The mystery was solved years later, in 2009. That’s when another scientist discovered that Trichoplax can sting an animal that tries to eat it. That sting can actually paralyze its would-be predator. It uses tiny dark balls, found on its upper side, to do this.
People had always thought those balls were just globs of fat. But instead, they hold some kind of venom that Trichoplax releases when attacked. In fact, the animal has genes that look a lot like the venom genes of certain poisonous snakes, such as the American copperhead and the West African carpet viper. A little blip of that venom means nothing to a big human. But if you’re a tiny snail, it can ruin your day.
Secret life
Pearse believes that scientists are still missing something big about Trichoplax. These animals usually reproduce by splitting in half. That gives rise to two animals. At least that’s what scientists see when they grow them in the laboratory. Once in a while, Pearse has seen one of these animals break into a dozen or more tiny pieces. Each would go on to become a new little animal.
But Trichoplax also reproduces sexually, as most other animals do. Here, a sperm — a male reproductive cell — seems to fertilize an egg cell from another individual. Scientists know this because they can find Trichoplax whose genes are a mix of two others. This suggests that the animal had a mother and father. Trichoplax also has genes that are involved in making sperm. Despite this genetic evidence of sex, says Pearse, “no one’s ever caught them at it.”
She also wonders if these animals have another life stage that no one knows about. Many sea animals, such as sponges and coral, start out as a tiny, baby larvae. Each larva swims around like a little tadpole. Only later does it land on a rock and grow into a sponge or a coral — one that will stay put for the rest of its life.
Trichoplax could also have a swimming larval stage. That larva’s body could look very different from the “sticky hairy plate” into which it later morphs. It also could help explain why such a simple-seeming animal has so many genes. Shaping and building that larval body would require many genetic instructions.
Pearse hopes that scientists can one day answer all of these questions. “These are mystery animals,” she says. “They have all kinds of puzzles waiting to be solved.”






