Stem cells can help build lab-grown organs that mimic real life
Such lab-made organoids can help us better understand disease and find new drugs
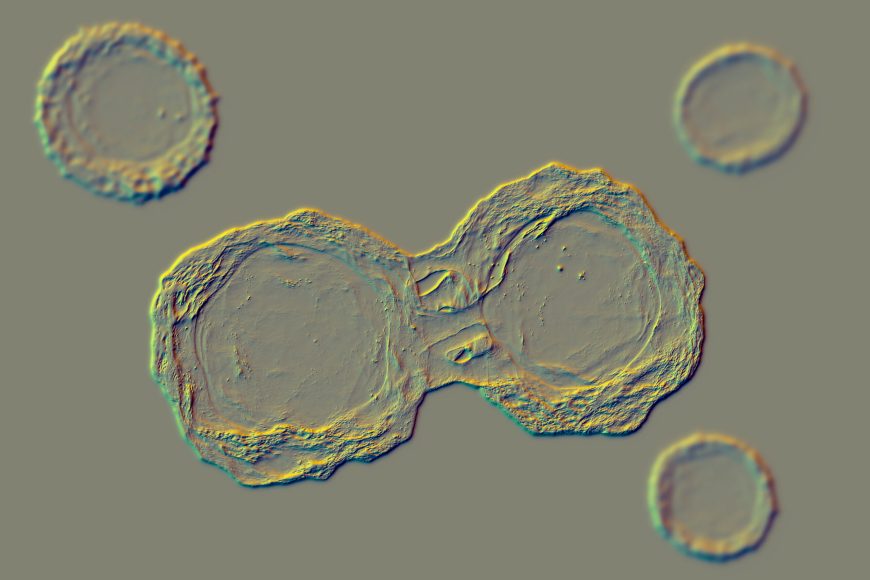
This picture illustrates dividing stem cells.
KATERYNA KON/SCIENCE PHOTO LIBRARY/Getty Images Plus
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
The organs inside our bodies team up to keep us alive. The heart pumps blood. Lungs pull in air so blood can deliver oxygen to cells. The liver filters blood and creates nutrients that keep us healthy. The liver also breaks down fat and removes poisons from the body. Scientists would like to study these tissues without harming them — or us. So they’ve been working to develop lab-made versions. But these mimics don’t work as well or perfectly copy the real thing. Plus, organs’ main mission is to collaborate, not to work in isolation.
Enter the organoid-builders, like Quinton Smith. A chemical engineer, he works at the University of California, Irvine. He and others are pioneering a new type of research. They’re marrying tech and biology to create better organ mimics. To answer scientific questions, these high-tech cellular communities can be poked, prodded, beamed at and hit with chemicals — so that our bodies don’t need to be.

To understand the role of most cells, “we study them in a petri dish,” says Quinton Smith. But a round, plastic dish does not resemble the cells’ normal environment.
In a petri dish, cells grow side by side in thin, flat layers. But inside people, those cells grow to form organs in a range of different shapes. Some are bulky. Others develop networks of tubes. Some evolve folds or layers that help filter what gets through them. Many spit out chemicals — hormones — that direct the actions of others. To keep us healthy, these organs must work together as a team.
It’s proven hard to recreate all that complexity in the lab. But researchers are merging biology and engineering to do just that. To make tiny electronics, electrical engineers create tiny circuits on computer chips, through a process called micropatterning. With 3-D printing, they can lay down the parts to build complex structures, sometimes with many different materials.
Similar printers are now beginning to construct complex biological structures. Some are like miniature, lab-grown organs. Known as organoids, they may also carry out some of the same roles of the organs they’re meant to mimic.
With organoids, Smith and others hope to study how diseases might affect internal organs. Organoids also can stand in for our organs to test the effects of potential drugs.
Tech made California’s Silicon Valley famous. By combining that tech with stem-cell biology, scientists at long last have begun “making tissues that look and react and function like human tissues,” Smith says. Until now, he says, “that hasn’t been done.”
The power of stem cells
Stem cells are cells that can change into any of many different specialized cells. One type of stem cell is proving useful in making organoids. Known as human pluripotent (Ploor-ee-POH-tunt) stem cells, these start out as normal body cells. Scientists then reset them to a very early stage of development.
That allows these cells to mature all over again, now into any type of needed cell. “It just blew my mind that you can take [pluripotent stem cells] and turn them into anything,” Smith says.
As a PhD student, he explored how physical and chemical cues can push these stem cells to become blood vessels. He was part of a team that used micropatterning. That’s where researchers stamp proteins onto glass slides. Those proteins help cells attach to the surface.
With micropatterning, the researchers helped stem cells organize into the beginnings of artificial blood vessels. Depending on the conditions, groups of cells could form 2-D stars, circles or triangles.
No tissue can grow without blood vessels. Oxygen is essential for cells to survive, and blood carries it to them. That why the ability to make vessels to carry blood or other liquids around the body is the “holy grail” of efforts to build organs in labs, says Milica Radisic. She’s a biomedical engineer in Canada. She works at the University of Toronto.
Another reason vessels are so important: They connect organs. “If I want to connect, let’s say, the heart and the liver to find heart-safe cancer drugs, I have to connect them to [vessels],” Radisic explains.

How to make a liver
Engineering can help researchers make liquid-carrying tubes in other organs, too.
Like branching blood vessels, a branching network of ducts carries bile acid throughout the liver and more. Bile is a greenish fluid that helps the body digest and absorb fat. But artificial liver tissue doesn’t always re-create bile ducts that branch as they do in the body.
To get around the problem, Smith and his team pour a stiff gel around tiny acupuncture needles. The needles create channels in the gel. Once the gel turns solid, the researchers plant stem cells inside. Then they douse the cells in a chemical that coaxes ducts to form. “We can create on-demand bile ducts using [this] engineering approach,” Smith reports.
Smith’s lab also uses 3-D printing. It ensures that liver tissues grown in the lab organize in the right way. While the cells inside our bodies can do this on their own, ones growing in the lab “need a little bit of help,” Smith explains.
Such engineering techniques can help researchers study and look for the root causes of some liver diseases. A notable one is called fatty liver disease. Hispanic people face a higher risk of this, Smith says. Comparing organoids grown from the cells of healthy people to those grown from cells of patients with liver disease may show why.
Looking beyond the liver
Organoids can help researchers study other organs, too. Smith and members of his lab, for instance, are starting to explore a disease that affects the placenta. It’s a flat organ that develops in the womb of pregnant people. It helps nourish the developing baby by supplying blood through an umbilical cord.
During pregnancy, some women develop a disease known as preeclampsia (PRE-eh-KLAMP-zee-uh). An inflammatory condition, it constricts the mother’s blood vessels. This can lead to dangerously high blood pressure. While any pregnant woman may develop this, African American women face a higher-than-normal risk of it.
Smith plans to examine organoid placentas. By studying them, he hopes to learn how physical forces and chemical cues from the organ affect the blood vessels attached to it.
“We’re really excited about this work,” Smith says. It’s only recently that scientists have tricked stem cells to enter an early enough stage of development to produce placentas. These lab-grown placentas even produce the hormone responsible for positive pregnancy tests: human chorionic gonadotropin (Kor-ee-AH-nik Guh-nad-oh-TROH-pin).
Chalk up yet another win for the power of stem cells.






