Magnets may one day cull deadly germs from blood
Tiny particles of antibody-covered iron would grab bacteria, then get mopped up by magnet
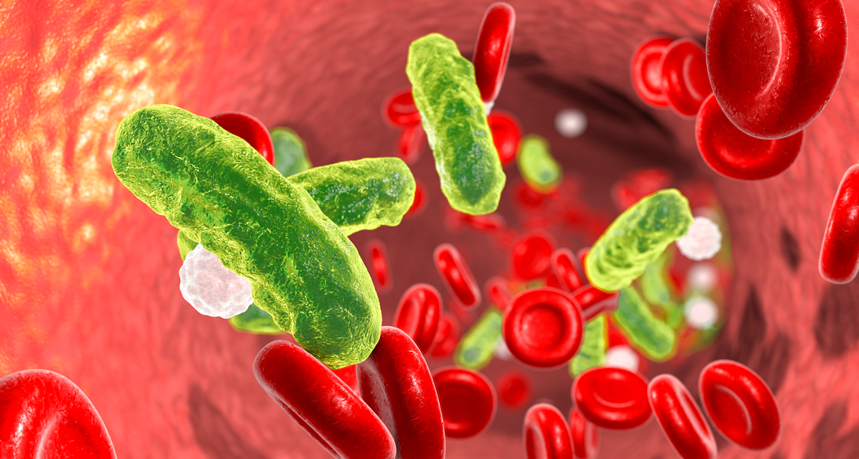
In sepsis, bacteria (colored green in this illustration) can cause a deadly infection. A newly proposed method might pull the invaders out of blood (red) with iron oxide and magnets.
Dr_Microbe/iStockphoto
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
By Sid Perkins
Sometimes the immune system overreacts to an infection. This mysterious condition is known as sepsis. Each year, more than a million people in the United States alone develop sepsis. And somewhere between a fourth and half of them will go on to die from it. But researchers have come up with a new tactic to attack these infections. They plan to filter germs from the blood. And it may only take simple tools: magnets and nano-bits of iron.
An infection anywhere in the body can trigger sepsis. Bacteria are usually to blame, although sometimes the source may be viruses or microscopic fungi. The worst cases tend to spread through the bloodstream. (That helps explain why sepsis is commonly, but wrongly, nicknamed “blood poisoning.”) Having so many possible causes makes sepsis tough to treat, says Gerald Pier. He’s a microbiologist at Brigham and Women’s Hospital in Boston, Mass. He also teaches nearby at Harvard Medical School.
Before doctors can treat sepsis, they must identify the germ responsible. So they will collect some blood and then send it to a lab. There, technicians multiply the microbes until there are enough to find them and identify them. That may take hours. And during that time, the sepsis may worsen dramatically. But Pier and some colleagues have an idea on how to start treating the bacteria even before they identify the germ.
The immune system uses antibodies to attack bacteria and viruses. These antibodies are proteins that latch onto the invaders as they’re carried around the body in blood. Like flags, antibodies make the microbes more visible to white blood cells. Then those white blood cells surround and consume the intruders.
Often, each antibody works against only one type of microbe, explains Pier. But his group developed a single antibody that is able to recognize nine of the 10 most common sepsis-causing bacteria.
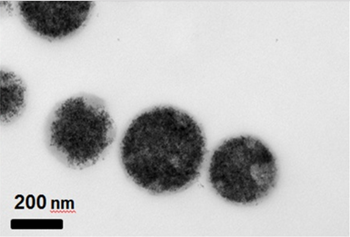
Still, antibodies alone aren’t enough. So others on the team invented a way to make the antibody stick to tiny particles of iron oxide. (Iron oxide is the scientific name for rust. And it is magnetic.) If those antibody-coated iron bits were mixed into a patient’s blood, the scientists reasoned, bacteria should stick to them. Then magnets could pull the iron particles from the blood — along with the bacteria that had glommed onto them.
Inge Katrin Herrmann is a chemical engineer at Empa in St. Gallen, Switzerland. (Empa is a short name for the Swiss Federal Laboratories for Materials Science and Technology.) The team that she and Pier are part of described their research in the November 28 Journal of Materials Chemistry B.
The researchers tested the concept with a blood-like solution.
Staphylococcus aureus bacteria are a major cause of sepsis. The researchers added different amounts of these germs to the liquid. Some solutions held just 100 S. aureus bacteria per milliliter. Others had 1,000 or 10,000 bacteria per milliliter. (For comparison, there are about 5 milliliters in a teaspoon.) Then the researchers added antibody-coated iron oxide particles to the solutions. The particles were about 300 nanometers (billionths of a meter) across. That’s smaller than most bacteria, which are usually a few micrometers (millionths of a meter) long.
After about 10 minutes, the antibody-coated nanoparticles had trapped at least 98 percent of the bacteria.
In the future, treating a patient would probably require using this germ-filtering strategy outside of the body, says Pier. Infected blood would be pumped through a machine. Magnets in the machine would remove the germs as they became coated with the iron-oxide and antibody mixture. The process would be a bit like dialysis, a treatment for severe kidney disease, explains Pier. In dialysis, a patient’s blood runs through a machine that removes wastes and extra water.

This filtering may sound complicated. But it would let doctors treat patients more quickly than current methods, says Herrmann. “We can remove what is in the bloodstream without having to identify it,” she says.
Besides removing bacteria from blood, the magnetic filtering would concentrate the bacteria. And that could speed up the process of identifying them, says Pier. Once doctors know what germ they’re dealing with. Then they can choose the proper drug to treat it.
Alan Cross is a doctor who specializes in infectious diseases at the University of Maryland School of Medicine in Baltimore. “This is an interesting concept, but it needs further testing,” he says. Magnetic germ removal still hasn’t been tested yet in animals or people, he notes. Such tests might reveal problems that don’t show up when using only cells or blood. For instance, the team’s tests suggest that bacteria attached to tiny iron particles stay alive. If these germs slipped by the magnets and reentered the body, they would likely migrate to the liver. (That organ is the body’s natural blood filter.) Once stuck there, the bacteria might grow and continue to foster infection.
Pier and Herrmann don’t think the germ-coated particles would get much chance to reenter the body. And even if they did, the researchers say, blood is slightly acidic. So it should soon dissolve the particles.
Cross also points to another potential problem. Treating a patient could take huge numbers of tiny iron particles. In the team’s tests, the researchers added about 10 billion particles to each milliliter of blood. But there are about 5 liters (1.3 gallons) of blood in the human body, notes Cross. So it would take about 50 trillion particles of iron to treat that much blood. “I’m astounded by the numbers,” he says.






