Nobel awarded for unveiling how cells recycle their trash
Winner used bread yeast to identify how cells break down — and then recycle — unneeded materials
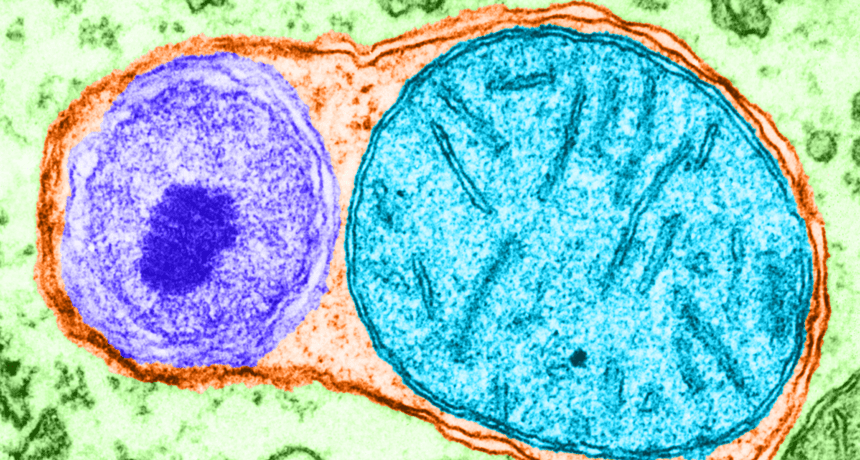
Cells fill pouches called vacuoles (orange) with cellular garbage (blue and purple) that they want to get rid of. This process of trash removal is called autophagy. The discovery of how it works just won the 2016 Nobel Prize in physiology or medicine.
Don W. Fawcett/Science Source, colorization by Mary Martin
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
By Meghan Rosen and Laurel Hamers
Healthy cells must do a good bit of housekeeping. If they didn’t, they soon would be clogged with old, broken or unneeded materials — what most people would think of as trash. Cells clean out their trash through a process that biologists call autophagy (Aw-TOF-uh-gee). But how it worked remained a mystery until a Japanese cell biologist did a lot of probing. What he uncovered won him the 2016 Nobel Prize for physiology or medicine.
The award was announced on October 3.
Cell biologist Yoshinori Ohsumi’s work falls into the category of physiology. That’s the science of understanding how biological parts work. His studies over the years, most recently at the Tokyo Institute of Technology in Japan, have focused on what happens to cells faced with starvation. Rather than give up and die, cells will begin to cannibalize parts they don’t need. Then they’ll recycle — eat or otherwise reuse — those unneeded parts.

But starvation is far from the only motivation for this autophagy, Ohsumi showed. Even healthy cells make use of this process to rid themselves of trash.
Too little recycling can cause cellular trash to build up. That can lead to brain disorders, such as Alzheimer’s and Parkinson’s diseases. Scientists have linked excess recycling, in contrast, with cancer.
“It’s so exciting that Ohsumi has received the Nobel Prize, which he no question deserved,” says Jennifer Lippincott-Schwartz. She’s a biologist with the Howard Hughes Medical Institute. She works at its Janelia Research Campus in Ashburn, Va. This year’s winner “set the framework for an entire new field in cell biology,” she says.
Indeed, Ohsumi‘s discoveries helped reveal the workings of a basic process that underlies biology, notes Maria Masucci. A biologist with the Karolinska Institute, near Stockholm, Sweden, she spoke during a news briefing on October 3. “There is growing hope,” she says, “that this knowledge will lead to the development of new strategies for the treatment of many human diseases.”
Tackling the largely unstudied process
Scientists got their first glimpse of autophagy in the 1960s. This was shortly after the discovery of a pouch within cells known as a lysosome (LY-soh-sohm). It acts as a garbage disposal, grinding fats and proteins and sugars into their basic building blocks. (This discovery would lead to a Nobel Prize in 1974 for Belgian scientist Christian de Duve.)
Researchers had observed lysosomes stuffed with big chunks of cellular wastes. But they also had observed another mystery pouch. Its role seemed to be that of a garbage truck taking those wastes to the lysosome for disposal.
Somehow, the cell had devised a way to consume large parts of itself. De Duve dubbed the process autophagy, from the Greek words for “self” and “to eat.” But for the next 30 years, how it worked remained pretty much a mystery.
“The machinery was unknown,” recalls Juleen Zierath, a Karolinska Institute physiologist. In fact, she notes, “How the system was working was unknown, and whether or not it was involved in disease was also unknown.”
That all changed in the 1990s. It was then that Ohsumi decided to study autophagy in a single-celled organism. He chose baker’s yeast, those microbes responsible for making bread rise. Studying the waste-treatment process in these cells was tricky. That’s partly because it happened so quickly. So Ohsumi bred special strains of yeast. They were not able to break down proteins in their cellular garbage disposals — pouches known as vacuoles (VAK-yoo-oles).
“He reasoned that if he could stop the degradation process, he could see an accumulation of the autophagy machinery in these cells,” Zierath explains.
And it worked.
When he starved these yeast cells, the “self-eating” machinery kicked into gear (presumably to scrounge up food for the cells). But because the garbage disposals were defective, the recycling machinery piled up in the vacuoles. In no time, they swelled like balloons stuffed with sand. Ohsumi could see the bulging, packed bags clearly under a microscope.
His team described this in a 1992 paper in the Journal of Cell Biology.
This opened a world of insights
The new technique let Ohsumi study the process in detail. Within about a year, he discovered as many as 15 genes needed for the cell-recycling machinery to work. Later, Ohsumi and others examined the proteins that these genes had instructed their host cells to make. From this information, the scientists began to figure out how the components of the “bulk waste” bag came together — and then fused with the lysosome.
The work revealed something new about the cell’s garbage centers, Zierath says. Before Ohsumi, biologists had assumed those lysosomes were little more than a trash dump, she notes. “But what he showed was that it wasn’t a waste dump. It was a recycling plant.”
Later, Ohsumi and his coworkers studied autophagy in animal cells and realized that the process played a key maintenance role in all kinds of cells — even those of mammals. Ohsumi “found a pathway that has its counterparts in all cells that have a nucleus,” says 2013 Nobel laureate Randy Schekman. He’s a cell biologist at the University of California, Berkeley. “Virtually every corner of the cell is touched by the autophagic process.”
(Story continues below image)
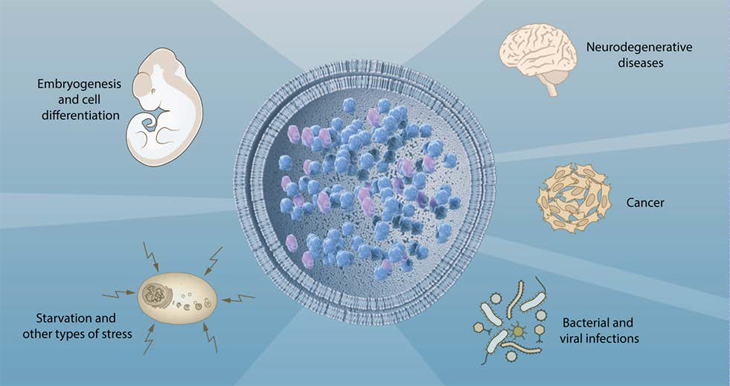
Since Ohsumi’s discoveries, research on autophagy has exploded, says Lippincott-Schwartz. “It’s an amazing system that every year becomes more and more fascinating.”
Ohsumi, 71, is still an active researcher. He received the call from the Nobel committee while he was at his lab in Japan. About his work, he said, “It was lucky. Yeast was a very good system, and autophagy was a very good topic.”
Still, he added in an interview with a Nobel representative, “we have so many questions. Even now we have more questions than when I started.”
Ohsumi will receive a medal and cash award of 8 million Swedish kronor (or about $934,000) at a December 10 ceremony in Stockholm. The award is named for Alfred Nobel. Best known as the inventor of dynamite, Nobel was a wealthy man when he died on December 10, 1896. In his will, he left much of his fortune to create prizes to those who have done their best for humanity in the fields of physics, chemistry, physiology or medicine, literature and peace.






