atom: The basic unit of a chemical element. Atoms are made up of a dense nucleus that contains positively charged protons and uncharged neutrons. The nucleus is orbited by a cloud of negatively charged electrons.
bond: (in chemistry) A semi-permanent attachment between atoms — or groups of atoms — in a molecule. It’s formed by an attractive force between the participating atoms. Once bonded, the atoms will work as a unit. To separate the component atoms, energy must be supplied to the molecule as heat or some other type of radiation.
cell: The smallest structural and functional unit of an organism. Typically too small to see with the unaided eye, it consists of a watery fluid surrounded by a membrane or wall. Depending on their size, animals are made of anywhere from thousands to trillions of cells. Most organisms, such as yeasts, molds, bacteria and some algae, are composed of only one cell.
cellulose: A type of fiber found in plant cell walls. It is formed by chains of glucose molecules.
chemical: A substance formed from two or more atoms that unite (bond) in a fixed proportion and structure. For example, water is a chemical made when two hydrogen atoms bond to one oxygen atom. Its chemical formula is H2O. Chemical also can be an adjective to describe properties of materials that are the result of various reactions between different compounds.
chemical bonds: Attractive forces between atoms that are strong enough to make the linked elements function as a single unit. Some of the attractive forces are weak, some are very strong. All bonds appear to link atoms through a sharing of — or an attempt to share — electrons.
chemical reaction: A process that involves the rearrangement of the molecules or structure of a substance, as opposed to a change in physical form (as from a solid to a gas).
DNA: (short for deoxyribonucleic acid) A long, double-stranded and spiral-shaped molecule inside most living cells that carries genetic instructions. It is built on a backbone of phosphorus, oxygen, and carbon atoms. In all living things, from plants and animals to microbes, these instructions tell cells which molecules to make.
electricity: A flow of charge, usually from the movement of negatively charged particles, called electrons.
fiber: Something whose shape resembles a thread or filament.
molecule: An electrically neutral group of atoms that represents the smallest possible amount of a chemical compound. Molecules can be made of single types of atoms or of different types. For example, the oxygen in the air is made of two oxygen atoms (O2), but water is made of two hydrogen atoms and one oxygen atom (H2O).
monomer: A molecule that is used as the basic building block of some larger molecule, known as polymers. From the Greek language, monomer means “one part.” (Polymer, also from Greek, means “many parts.”)
plastic: Any of a series of materials that are easily deformable; or synthetic materials that have been made from polymers (long strings of some building-block molecule) that tend to be lightweight, inexpensive and resistant to degradation.
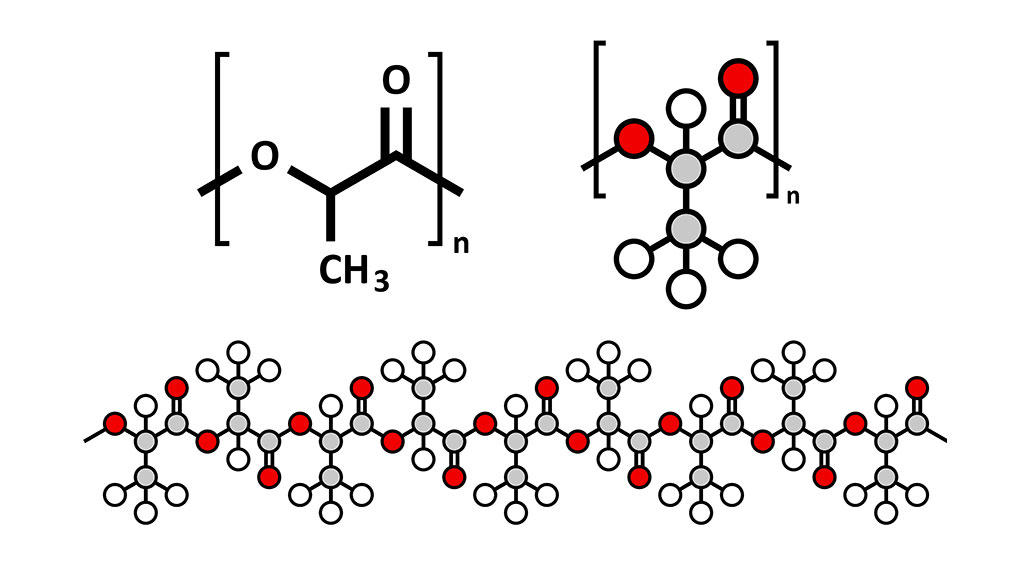
polymer: A substance made from long chains of repeating groups of atoms. Manufactured polymers include nylon, polyvinyl chloride (better known as PVC) and many types of plastics. Natural polymers include rubber, silk and cellulose (found in plants and used to make paper, for example).
protein: A compound made from one or more long chains of amino acids. Proteins are an essential part of all living organisms. They form the basis of living cells, muscle and tissues; they also do the work inside of cells. Among the better-known, stand-alone proteins are the hemoglobin (in blood) and the antibodies (also in blood) that attempt to fight infections. Medicines frequently work by latching onto proteins.