New nano-cages snag and hold gases
A tiny holding cell can capture and retain hard-to-snatch gases
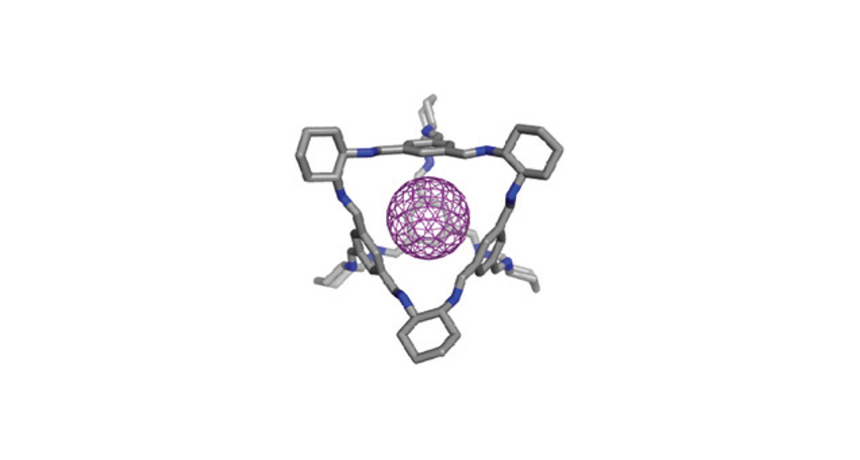
The purplish blob in the middle is an atom of a noble gas. The surrounding blue and gray structure represents a cage that holds the atom for a while. This artist's drawing represents a new kind of molecular cage built by scientists. Such a device could help trap harmful chemicals in the air.
L. CHEN ET AL/NATURE MATERIALS
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
Trapping an individual atom requires a tiny cage. Scientists recently stumbled on a way to make just such a chemical trap. Each of these nano-scale holding cells can snag and hold atoms of radon, krypton and xenon. All three elements are noble gases — and each is notoriously difficult to catch.
Noble gases have no smell, taste or color. They also keep to themselves, which means they don’t ordinarily interact with other elements. But these elements are important — and sometimes potentially dangerous. Studies have linked breathing high levels of naturally occurring radon, for instance, to lung cancer. Krypton and xenon dazzle with color when they’re electrically charged, so both are used in glowing signs and lasers. But nuclear power plants produce forms of krypton and xenon that could be dangerous to people.
Andrew Cooper is a materials scientist at the University of Liverpool in England. In the lab, he and his coworkers created a synthetic — non-natural — molecule that acts like a cage. It can help separate individual atoms. The scientists who developed the synthetic substance hope these special molecular cages might find use in monitoring the air in homes or jailing runaway atoms of these rare elements.
Cooper’s group described its new noble-gas cage July 20 in Nature Materials.
Other scientists find the new achievement quite exciting. “I would die to have these compounds in my hands,” Siegfried Waldvogel told Science News. An organic chemist, he designs chemical sensors at the Johannes Gutenberg University Mainz in Germany
In fact, finding the new cage was a “happy accident,” Cooper told Science News.
He and his colleagues had been trying to make a polymer, which is a material made of identical building blocks that have been linked into a long, chainlike structure. (Plastic is an example of a polymer.) In some polymers, these repeated building blocks are organic. In chemistry, “organic” refers to compounds that include the element carbon.
But Cooper’s group didn’t produce a polymer. It ended up instead making clumps of tiny, three-dimensional cages. Each cage was made from organic molecules.
The inside of each cage is about 0.44 nanometer across. (A nanometer is one-billionth of a meter.) That’s the right size to hold atoms of krypton, xenon and radon. Each is between 0.37 and 0.42 nanometer in diameter.
Getting atoms into the cage might be the trickiest part. The entrance to the cage is only 0.36 nanometer across. That’s smaller than any of these atoms. However, all organic molecules wiggle just a bit. Cooper and his colleagues say the chemical “bars” of their cages wiggle enough to let the atoms in or out.
The scientists tested their cages by blowing a mix of common elements plus xenon or krypton at the cages for 45 minutes. Elements like nitrogen, oxygen and carbon passed right through each molecular cage. But the cages trapped xenon for about 15 minutes. Krypton paused in the cage only briefly, but the scientists say even a short stay is enough to snatch the atoms from the air.
Xenon, useful in medical devices and lighting, sells for about $5,000 per kilogram (2.2 pounds). The new cages could assist in the harvest of this valuable gas. Or they could be used in a filter to snag toxic noble gases from the air so that they don’t pose health risks.
Power Words
atom The basic unit of a chemical element. Atoms are made up of a dense nucleus that contains positively charged protons and neutrally charged neutrons. The nucleus is orbited by a cloud of negatively charged electrons.
cancer Any of more than 100 different diseases, each characterized by the rapid, uncontrolled growth of abnormal cells. The development and growth of cancers, also known as malignancies, can lead to tumors, pain and death.
chemical A substance formed from two or more atoms that unite (become bonded together) in a fixed proportion and structure. For example, water is a chemical made of two hydrogen atoms bonded to one oxygen atom. Its chemical symbol is H2O.
chemistry The field of science that deals with the composition, structure and properties of substances and how they interact with one another. Chemists use this knowledge to study unfamiliar substances, to reproduce large quantities of useful substances or to design and create new and useful substances.
element (in chemistry) Each of more than one hundred substances for which the smallest unit of each is a single atom. Examples include hydrogen, oxygen, carbon, lithium and uranium.
krypton An inert gas that’s a member of the noble gases, used in some types of electric lights.
molecule An electrically neutral group of atoms that represents the smallest possible amount of a chemical compound. Molecules can be made of single types of atoms or of different types. For example, the oxygen in the air is made of two oxygen atoms (O2), but water is made of two hydrogen atoms and one oxygen atom (H2O).
nano A prefix indicating a billionth. In the metric system of measurements, it’s often used as an abbreviation to refer to objects that are a billionth of a meter long or in diameter.
noble gas Any of the elements helium, neon, argon, krypton, xenon and radon, all of which exist naturally as gases.
nuclear power Energy derived from processes that produce heat by splitting apart the nuclei of atoms (fission) or forcing atomic nuclei to merge (fusion). A nuclear power plant uses that heat to drive turbines that create electricity.
organic (in chemistry) An adjective that indicates something is carbon-containing; a term that relates to the chemicals that make up living organisms.
polymer Substances whose molecules are made of long chains of repeating groups of atoms. Manufactured polymers include nylon, polyvinyl chloride (better known as PVC) and many types of plastics. Natural polymers include rubber, silk and cellulose (found in plants and used to make paper, for example).
radioactive An adjective that describes unstable elements, such as certain forms of uranium and plutonium. Such elements are said to be unstable because their nucleus sheds energy that is carried away by photons and/or and often one or more nuclear particles. This emission of energy is by a process known as radioactive decay.
radon A radioactive gas belonging to the noble gases. It is naturally given off by some rocks in the soil. When it accumulates in homes or water supplies, it can pose a health hazard.
synthetic (as in materials) Materials created by people. Many have been developed to stand in for natural materials, such as synthetic rubber, synthetic diamond or a synthetic hormone. Some may even have the same chemical structure as the original.
xenon An inert gas that’s a member of the noble gases. It’s used in some kinds of electric light.







