Why some icicles become scallops not spikes
The newfound quirky shapes reflect the surrounding water’s density
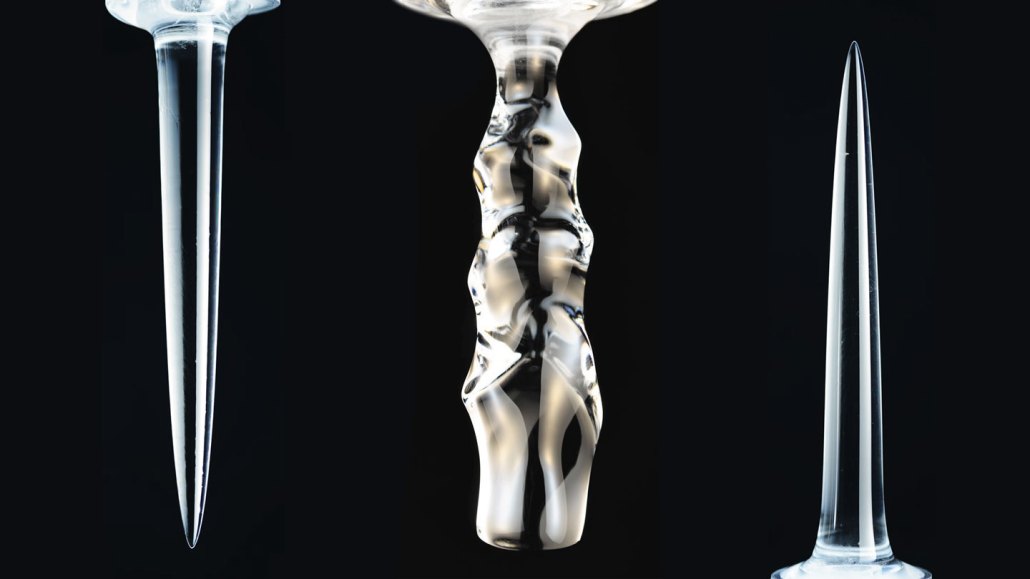
When columns of freshwater ice were submerged in water, they melted. But depending on the water’s temperature — and density — the icicles that formed could be downward-facing spikes (left), scallops (center) or up-facing spears (right).
Applied Math Lab/NYU
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
Water’s density can have wacky impacts that researchers are still discovering. New experiments show, for instance, that ice columns submerged in liquid water can melt into three distinct shapes. Which they become will depend on the water’s temperature.
Leif Ristroph is a mathematician in New York City. He was part of a team at New York University that anchored in place cylinders of ultrapure ice. Each cylinder was up to 30 centimeters (11.8 inches) long. Typically, liquids become denser the cooler they get. But freshwater is densest at 4° Celsius (39.2° Fahrenheit). At lower temps, the water becomes less dense. To test how that might affect the melting ice, the researchers submerged their ice cylinders in water between 2 °C and 10 °C (35.6 °F and 50 °F).
And “almost everything” about the experiments was surprising, Ristroph now reports.
The ice melted into smooth, downward-pointing spikes when placed in water cooler than about 5 °C (41 °F). When the researchers used a computer to model this behavior, it showed “a strange thing,” Ristroph says. “The cold liquid water near the ice is actually buoyant.” It wants to float because it is less dense than the rest of the water in the tank. That upward flow of melt water draws warmer water closer to the ice’s base. And this causes ice near the bottom to melt faster than at the top.
The opposite occurred when the water was warmer than about 7 °C (44.6 °F). Now the ice formed an upward-pointing spike. The colder water near the ice is now denser than the body of water around it. So that colder water sinks. This pulls in warmer water at the top of the ice, causing it to melt faster than at the bottom, the computer model explains.
Between about 5 °C and 7 °C, the ice melted into scalloped columns. “Basically, the water is confused,” Ristroph says. It ends up forming layers of different densities. Some layers tend to rise as others sink. In the end, the water organizes itself into “swirls or vortices of fluid.” And this carves “weird ripples into the ice,” he observes.
His team shared its new findings January 28 in Physical Review Letters.
Not the end of the story
“Our applied-math lab studies fluid dynamics,” Ristroph says. This work, he says, was its “first little dip into ice.” More work is needed to understand the complex mix of factors that can trigger the varying shapes that form as ice melts in nature.
The water in these tests, he notes, was “all quiet” when the experiments began. But in nature, motion can be created by all sorts of things — swimming animals, boats full of people, cargo and more.
And next, Ristroph plans to study what happens to the ice when the saltiness — or salinity — of seawater varies along with temperature.
He’s also started looking at what happens when ice cylinders are put in the tanks without anchoring them in place. Such free ice, Ristroph points out, can “move or flip over.” His team is curious how all of these movements may affect the shapes that melting ice can take on — whether as cubes in your glass or icebergs on the high seas.
The study of ice contains “a beautiful set of problems,” Ristroph says. “I think you could spend a whole career just on ice and never run out of stuff to do.”




