Will bacterial ‘wires’ one day power your phone?
These protein strings can pull electricity out of moist air anywhere

Nanowires from bacteria can turn water into electricity.
Ella Maru Studio/Courtesy of UMass Amherst
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
When Jun Yao started a new job, this electrical engineer wasn’t looking to create a “green” source of power. But chance helped him discover a way to use all-natural protein to turn water into electricity.
Yao works at the University of Massachusetts (UMass) in Amherst. He uses nanowires in the electronics he’s been designing. These wires are super tiny, each just a billionth of a meter (three-billionths of a foot) wide. But as a new professor, Yao had a hard time getting enough of them for his research.
Discouraged, he mentioned the problem to Derek Lovley. This microbiologist also works at UMass. Lovley told Yao about bacteria that make nanostrands of protein. To find out if these might substitute for nanowires, the pair teamed up.
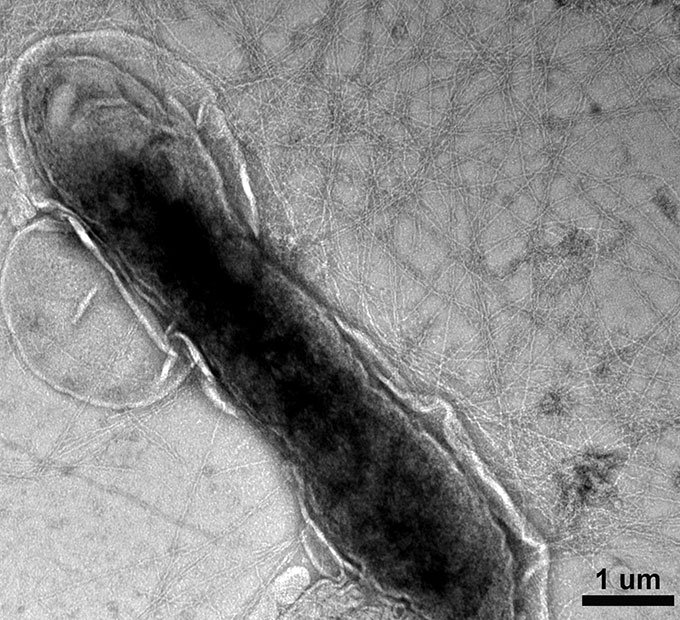
Geobacter bacteria live in mud. Lovley first discovered these microbes more than 30 years ago. Since then, these microbes have been used to clean up oil spills and radioactive waste.
The bacteria grow wire-like protein strands all over the outside of their cells. “They look like a miniaturized sea urchin,” Yao says. As the microbes turn food into energy, they release electrons. Those electrons move through the protein strands, ending up on iron in the mud.
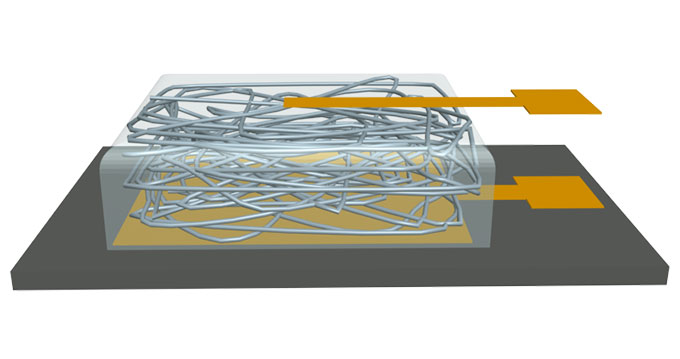
For the new research, Lovley removed the nanostrands from billions of these bacteria. Yao’s team then sandwiched a cloud of the wirelike strands between two small, gold metal plates. (Imagine taking a handful of threads and smooshing them flat.) The gold serves as electrodes. They make contact with the non-metal part of an electric circuit (those protein wires). Graduate student Xiaomeng Liu then applied a voltage between the two electrodes. Yao likens this to connecting them to a battery. When Liu did this, electricity — the flow of electrons — moved through the system. The protein “wires” now behaved like metal ones.
Happy accident
One day, Liu forgot to turn on the voltage. Yet he saw electricity was flowing through the device anyway. To his surprise, the protein nanowires had created electricity. After testing, the researchers showed that the air’s humidity — water content — had been powering the setup.
Excited, the researchers decided to test how well their new system worked. They started with one tiny device. Its bottom electrode was just 5 millimeters (0.2 inch) on a side. Atop this was a layer of nanowires 7-micrometers thick (that’s much thinner than a human hair). On top sat a smaller, square electrode, 1 millimeter on each side.
The device produced electricity at all humidity levels tested, but it made more when humidity was high. At top power, it produced a sustained 0.5 volt. When the researchers connected five devices, they got five times the energy output. Covering the device to keep water out of the nanowires shut off its electricity-making. Removing the cover turned the device on again. Though the output of a single device is tiny, a group of them could charge a phone or light a lamp, Yao says.
Key to the system are small spaces between the nanowires, called nanopores. They allow water to move between the wires. More water collects on the side with the small electrode where the nanowire package contacts the air. Less gathers on the side where the nanowires touch the larger electrode. This difference, or gradient, makes a positive charge build up on one side of the “wires” and a negative charge on the other. It’s a bit like how lightning forms, Yao says. “The movement of water molecules creates a charge separation in the cloud,” he explains. “Eventually it reaches a threshold so the cloud discharges,” producing lightning.
The team described its invention February 27 in Nature.
Powering the future?
The new device has the potential to be a major innovation in renewable energy, Yao says. After all, he notes, “humidity is everywhere.” The devices are very thin and can be stacked. Unlike solar panels, they don’t need light or to cover a big area. They can be used indoors or out. The can even become part of furniture, cell phones and more without being noticeable.
The best part, Yao says, is that harvesting the microbial wires produces no harmful chemicals. And when the devices are no longer needed, the gold electrodes can be reused or recycled. The nanowires can be tossed out, allowing the protein to break down naturally. This means that unlike other types of renewable energy, Yao says, there’s no long-term waste to pollute the environment.
This looks to be an important technology, says Quanbin Dai. He’s a nanotechnology researcher who was not involved with the study. He works at Case Western Reserve University in Cleveland, Ohio. Many people have “cell phones and wearable electronics that need to be recharged,” he notes. The idea of being able to power these from humid air is appealing, he says. Protein nanowires could make electric power anywhere and at any time of day. “It will be interesting to see the successful implementation of this,” he says.
Yao and Lovley are already working to make that happen. One limit right now is how to grow enough microbial nanowires quickly. But Lovley is on it. He already has inserted the gene for making nanowires into a faster-growing type of bacteria.
This is one in a series presenting news on technology and innovation, made possible with generous support from the Lemelson Foundation.







