Reversible superglue mimics snail slime
This super-strong adhesive can be undone by adding water

When not on the move, snails use mucus to stick to plants and other objects.
Gabriel Pacce/iStock/Getty Images Plus
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
Makers like to combine unusual items to create new things. To do so, they need super-strong adhesives — tapes and glues — to hold it all together. But sometimes they want to be able to take the items apart again. That’s been a problem, because reversible adhesives usually are not very strong. Sticky stuff can be super-strong and permanent — like superglue. Or it can be less sticky but easily removed — think of a sticky note. Now, though, researchers have created an adhesive that’s both reusable and super-strong.
Shu Yang works at the University of Pennsylvania in Philadelphia. Her team described their new superglue July 9 in the Proceedings of the National Academy of Sciences.
As a materials scientist, Yang uses physics, chemistry and engineering to create new types of items. In her work, Yang often finds inspiration for new materials based on structures that exist in nature.
For years, she has been working to create an adhesive that not only holds well but also can be undone and reused. Earlier work mimicked the tiny hairs on gecko feet. Although the material came unstuck easily, it didn’t have a strong grip. So the scientists in Yang’s lab kept searching for something better.
One day, a student in her lab was playing with a substance known as a hydrogel. A polymer, it’s made up of repeating chains of smaller chemicals. This particular gel turns soft when wet — in fact, it’s what makes contact lenses so flexible. Yang’s lab had been using it to make various structures for about 10 years. The student, Gaoxiang Wu, made patterns with it on a glass slide and then left it there.
When Wu returned, the hydrogel had hardened and was seriously stuck. He pulled, pried and scraped, but nothing separated the gel from the glass slide. Then he added water — and it came right off.
That finding made Yang and her team curious. Why had this dried gel been so hard to remove? They also wondered whether anything in nature might also work that way. And before long they discovered that snails make a similarly sticky goo.
During the heat of the day, snails are at risk of drying out. To prevent this, a snail finds a good spot near the ground with plenty of moisture. There, it pumps lots of mucus through the opening on its shell.
The mucus oozes over the ground, filling in any gaps. As it dries, the mucus hardens. This creates a structure that is both protective and adhesive. Called an epiphragm (EP-ih-fram), it seals the moist snail inside its shell, protecting it from predators that would readily munch on it if they could get to the meat inside. When temperatures fall in the evening and humidity rises, the mucus loosens. Now free to move, the snail continues on its way.
A sticky situation
Yang and her team reached out to Anand Jagota. He is a bioengineer at Lehigh University in Bethlehem, Penn. Jagota specializes in soft materials and their adhesive properties. Together, the researchers studied the hydrogel and found that it worked the same way as a snail’s slime. When the gel was wet, it oozed into every little nook and cranny, just like snail mucus. When it dried, the material turned hard and glassy. Now it was almost impossible to pull off.
The key, Yang says, is that the gel is very soft when wet. It’s a lot like those squishy splat balls that stick to the wall, she says. Almost every surface has tiny imperfections, she observes. So with just a small bit of pressure, the gel can squeeze into all of those the tiny pores and spaces on the surface it’s sticking to.
For most adhesives, that surface roughness is a problem. It reduces the amount of contact between the adhesive and surface, she notes. That makes the adhesive less sticky. But the hydrogel fills in those itty-bitty gaps, forming a tight connection.
What’s more, the gel doesn’t shrink much as it dries. So it doesn’t pull away from those rough patches. Instead, it holds its shape as it turns glassy. In fact, that’s key to why it sticks so well — it’s locked on to the tiny cavities. “We couldn’t separate the gel from the substrate,” Yang says. No matter what tool they tried.
Hanging tight
Strong adhesives are important for making things — building cars or airplanes, for example. So the team wanted to figure out just how strong their new adhesive was.
To find out, they made the gel on one side of a sheet of silicon. They cut the gel-coated silicon into small chips no bigger than a postage stamp. Then they superglued a piece onto each side of a strip of heavy-duty metal. They glued other squares of bare silicon to one end of each of two Kevlar ribbons. (Kevlar is a tough material used to make bulletproof vests. It wouldn’t fall apart during testing.)
Finally, the team added water to the hydrogel and pressed the ribbons’ bare silicon wafers onto either side of the gel-coated metal strip. When it dried, the Kevlar ribbons were firmly attached to the metal.
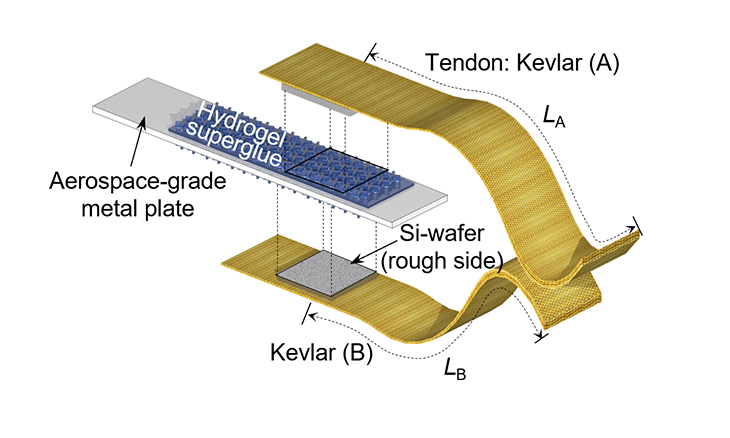
The team used the hydrogel to attach the metal strip to the front of a filing cabinet. They then hung a 4-kilogram (8.8-pound) weight from the Kevlar ribbon. It held firmly. Only when the team added water did the hydrogel soften, letting the weight fall to the ground.
The hydrogel was even stronger than the superglue. Sometimes when the weight was added, the superglue used in the setup broke free. But the hydrogel always held fast.

In a final test, the team built a large metal frame and stuck the gel-coated strip with the Kevlar ribbons to the top. They attached the Kevlar to a harness worn by a student in the lab. He carefully lifted his feet off the ground — and waited. The strip held. Just two postage-stamp-sized pieces of gel held up an 87-kilogram (192-pound) man!
The new adhesive “beautifully imitat[es] the reversible bonding mechanism used in the snail’s epiphragm,” says Diana Kay Hohl. She is a materials scientist at the University of Fribourg in Switzerland. Water is a good choice to make the new adhesive non-toxic, she says. But water is slow to dry. The hydrogel takes longer to dry than many other strong glues, Hohl notes. And water won’t work in many types of manufacturing, she points out.
“It will be interesting to see if, in next-generation materials, the adhesion can be tuned by triggers other than water,” she says. Heat or light, for example, might make the super-adhesive more suitable for manufacturing.
This is one in a series presenting news on technology and innovation, made possible with generous support from the Lemelson Foundation.






