The newest elements finally have names
These superheavies honor the places or people instrumental in the discovery of such rare elements
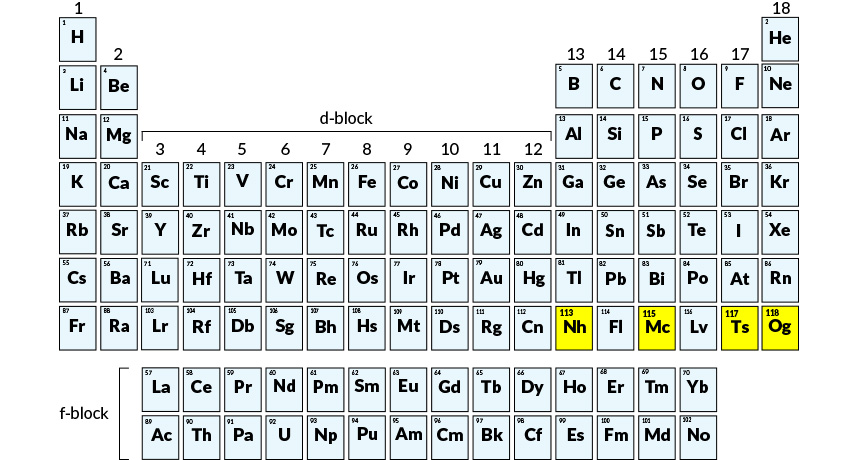
Four elements, officially added to the periodic table in December (and highlighted here in yellow), now have names that honor Japan, Moscow, Tennessee and a Russian physicist.
E. Otwell
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
On December 30, the International Union of Pure and Applied Chemistry, or IUPAC, announced the official discovery of four new elements. But back in December, none of these newbies yet had a name. That had to wait until today.
Elements 113, 115, 117 and 118 — fill out the seventh row of the periodic table of the elements. All are superheavies. That’s why they sit at the bottom right of the table (see above).
Naming rights typically go to those who discover an element. And that’s what happened here. Element 113 was discovered by scientists at RIKEN in Wako, Japan. They’ve asked to call it nihonium, to be abbreviated as Nh. This name comes from Nihon. It’s Japanese for “Land of the Rising Sun,” which is what many people call Japan.
Element 115 will become moscovium, shortened as Mc. It refers to the Moscow region. And that was where the Joint Institute for Nuclear Research is based (Dubna). It discovered number 115 in collaboration with researchers at Lawrence Livermore National Laboratory in California and Oak Ridge National Laboratory (ORNL) in Tennessee.
Tennessee also gets a periodic table shout-out. It’s the home state of ORNL, Vanderbilt University and the University of Tennesse. So element 117 will become tennessine. It will bear the symbol Ts.
Russian physicist Yuri Oganessian was involved in the discovery of several superheavy elements. So the group behind number 118 decided to name it after him. It becomes oganesson — or Og.
“I see it as thrilling to recognize that international collaborations were at the core of these discoveries,” says Jan Reedijk at the Leiden Institute of Chemistry in the Netherlands. He contacted the labs involved with the newly discovered elements and invited their scientists to propose names for them. Those names, Reedijk says, now “make the discoveries somewhat tangible,” meaning seemingly more real.
Element names have to follow certain rules. So silly choices such Element McElementface would not be accepted. What are allowed: names reflecting a scientist, a place or geographic location, a mineral, a mythological character or concept, or some feature that is characteristic of the element.
The newly recommended names are now open to review by IUPAC and the public through November 8. After that, the names will be official.
And that’s not the end of activities to tweak the periodic table. Physicists already are probing for even heavier elements. These would sit in a new eighth row on the table. Some scientists also are working to confirm that copernicium is real. Somewhat smaller than the newest elements, it would be number 112.
To evaluate all of this ongoing work, chemists and physicists are about to set up a new group. They will review claims of any additional new elements.






