Recycling a climate-warming gas could make ‘greener’ farmed fish
Instead of warming the climate, methane can help farmers — and may save some fish, too
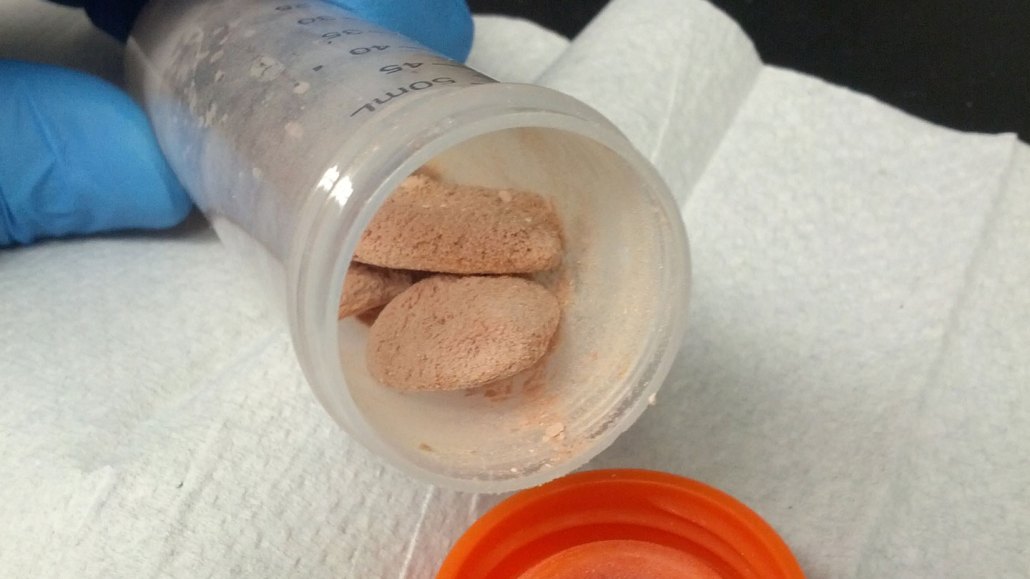
Researchers proposed a low-cost way to turn methane into fish feed, such as the pellets shown here.
Jaewook Myung
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
Some 3 billion people today rely on fish as a high-quality source of protein. Much of that fish is now “farmed,” which can be hard on the environment. But researchers think they have a way to make farmed fish a bit “greener.” Their technique could also help deal with a second problem — growing releases of methane, a climate-warming gas. Their proposal: Use that greenhouse gas to make food for fish or other livestock.
Oil and natural gas companies, wastewater-treatment plants, landfills and livestock all generate lots of methane pollution. As a greenhouse gas, methane absorbs the sun’s heat, and quite well. Over a century, a kilogram of methane will warm the atmosphere 25 times more than a kilogram of carbon dioxide (or CO2).
Fortunately, methods exist to capture methane effectively, says Sahar El Abbadi. She’s an environmental engineer at Stanford University in California. She also led the new study. Most companies don’t capture methane wastes. El Abbadi says they find it easier to just flare the gas — that is, burn it. (That burning changes methane into CO2, which creates a new problem.)
When El Abbadi joined Stanford as a PhD student, her supervisor was making a new type of plastic. Bacteria could break it down so that it would not become a long-lived pollutant. But the plastic’s source was crude oil. That fossil fuel was neither renewable nor clean.
Around the same time, researchers in Belgium discovered that fish and shrimp would happily dine on bacteria that degrade this new plastic. This intrigued El Abbadi. She wanted to find new uses for wastes from the oil and mining industries. Among those wastes: flared natural gas.
In her research, El Abbadi learned of companies that are using methane to make food-grade proteins. She wondered if she could feed fish this protein. And here’s why that could help the environment. Today, most farmed fish are fed fish meal. That’s dried, ground-up fish that might otherwise be eaten by people or other animals.
One problem: People making that protein were buying natural gas to get their methane. This made those new proteins costly. El Abbadi’s team now proposes getting bacteria to eat the methane, and then feed those protein-rich microbes to the fish. These proteins might also be fed to other types of farmed animals.
Growing the bacteria
The Stanford researchers’ first step was finding microbes to eat that methane. These are known as methanotrophs (Meh-THAN-oh-troafs). They are very common, notes Marina Kalyuzhnaya. She works at San Diego State University in California, where she studies such methane-eaters. These microbes “can be collected from lakes, soil, the ocean, rivers and even plant roots,” she explains.
To grow those bacteria in the lab, El Abbadi used a special container. Then her team filled the container halfway with cold water and added the methane-hungry microbes. One pipe entering the container supplied a stream of methane. Additional pipes brought in other gases the microbes needed to grow, breathe and make proteins.

A population of methanotrophs will double every three hours, Kalyuzhnaya says. This means, if you place 28 grams (an ounce) of the microbes in the container and keep feeding them methane and nutrients for 24 hours, you can end up with more than 7 kilograms (15.4 pounds) of the bacteria!
Every so often, this growing bounty of bacteria would be filtered out, dried and processed as fish food. El Abbadi’s team described its new process November 22 in Nature Sustainability.
New protein source could be affordable
By focusing on waste methane, the new process could become quite affordable, the Stanford team says. Electricity to maintain the temperature of the microbe-growing chamber is the only big expense. Where the price of electricity is low, a ton of fish feed can be produced for $386 less than the cost of most fish meals, the researchers say.
And if they find methane-eating bacteria that grow in far warmer conditions, El Abbadi says, “then the price of [this] fish feed can get even cheaper.”
This new system “is mimicking the food cycle found in nature,” observes Dorian Leger. He works in Germany at the start-up firm Connectomix. (It’s a biotech company.) Last summer, he was part of a team that studied an efficient way to make similar types of bacterial proteins. Instead of methane, however, his team used energy from solar panels to alter CO2 so that it could be used in producing proteins.
“The Stanford technique has incredible potential,” says Kalyuzhnaya. But it should really be performed on a large scale, she notes. At a small scale, she explains, it could be too much work and use too much energy.
Correction: This story was changed to denote Dorian Leger’s new affiliation.






