Steady heartbeats may depend on white blood cells
Known as macrophages, those blood cells had been thought to just fight germs

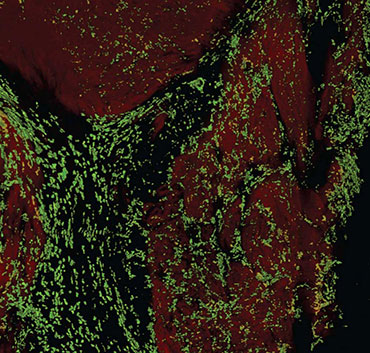
Like jumper cables, macrophages (green) “plug in” to heart cells (light purple and pink), providing an electrical boost. It keeps them beating on time, pumping blood around the body, a study in mice shows.
ELLA MARU STUDIO
Share this:
- Share via email (Opens in new window) Email
- Share on Facebook (Opens in new window) Facebook
- Share on X (Opens in new window) X
- Share on Pinterest (Opens in new window) Pinterest
- Share on Reddit (Opens in new window) Reddit
- Share to Google Classroom (Opens in new window) Google Classroom
- Print (Opens in new window) Print
Immune system cells are supposed to fight disease. But some may also play an everyday role in helping your heart beat on time. That’s the unexpected finding of a new study.
The immune cells are a type of white blood cells known as macrophages (MAK-roh-FAY-zhuz). They usually protect the body from invading disease-causing microbes. But in mice, at least, they also help electricity flow between heart-muscle cells.
A steady pattern of electrical zaps instruct the heart’s muscle cells each time it should contract. This creates their rhythmic pattern, known as the heartbeat. Those contractions pump blood through the heart and on throughout the rest of the body.
Macrophages squeeze in between heart muscle cells. There, they “plug in” to the muscle cells. Doing so aids the heart cells in receiving the electrical signals they need to stay on beat.
Matthias Nahrendorf is a cell biologist at Harvard Medical School in Boston, Mass. He and his team shared this new discovery April 20 in the journal Cell.
Electrifying find was an accident
Researchers have known for several years that macrophages live in healthy heart tissues. But what they did there was “still very much a mystery,” says Edward Thorp. He’s an immune-system expert at Northwestern University’s Feinberg School of Medicine in Chicago, Ill. He was not involved in the new research.
Nahrendorf was curious about those macrophages in the heart. So he tried to perform an MRI scan on the heart of a mouse that was genetically engineered to not have macrophages in their hearts. The scan wasn’t successful. The animal’s heartbeat was too slow and irregular to get a scan.
Those symptoms pointed to where the heartbeat problem likely was. It’s a bundle of muscle fibers known as the atrioventricular (AY-tree-oh-ven-TRIK-u-lur) node. This AV node electrically connects the upper and lower chambers of the heart.
People with AV-node irregularities may need a pacemaker to keep their heart beating as it should. In healthy mice, researchers discovered macrophages concentrated in the AV node. What the cells might be doing there, however, had been unclear.
So Nahrendorf’s team isolated a single heart macrophage and then tested it. It showed no electrical activity. As a result, the mystery remained. Then the researchers linked a macrophage to a heart-muscle cell. The two began communicating electrically. Now an answer to that mystery started to emerge. After all, electrical messages are what stimulate heart-muscle cells to contract.
Chemistry plays a role
Ions are charged molecules. And heart muscle cells have an imbalance of them. While resting, there are more positive ions outside a heart-muscle cell than inside it. When a muscle cell receives an electrical signal from a neighboring heart cell, that distribution of ions switches. Now there are more positive ions inside the cell than outside it. That switch is called a depolarization (De-POH-lur-ih-ZAY-shun).
This brief change causes the cell to contract. And this sends the electrical signal on to tell the neighboring muscle cell to depolarize.
Scientists previously thought that heart-muscle cells could make this ionic shift on their own. But Nahrendorf’s team found that macrophages play a role, too. Using a protein, a macrophage hooks onto a heart-muscle cell. This protein directly connects the inside of these cells to each other. Doing so lets macrophages transfer positive ions — and a positive electric charge. It’s like using that protein as a jumper cable to give the muscle cell a little electrical jumpstart. This makes it easier for the heart cells to depolarize and trigger the heart contraction, Nahrendorf says.
“With the help of the macrophages, the conduction system becomes more reliable,” he says. That means, he adds, “it is able to conduct faster.”
Nahrendorf and his colleagues have found macrophages within the AV node in human hearts, too. They don’t know, however, if these cells play the same role in people. The next step will be to confirm their role. The team also wants to explore whether heart problems such as arrhythmias might be due in part to an absence of macrophages. Or maybe the cells are no longer so good at jumpstarting heart-muscle cells any more.
Thorp at Northwestern calls the study’s conclusion that macrophages electrically work with heart-muscle cells as “paradigm shifting.” By that he means, they could alter our basic understanding of how these cells work. Indeed, Thorp says, the new data highlight the importance of macrophages, beyond their role in defending the body from germs.






